 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

|
Planck and the Quanta |
|
|
Planck's constant and the Energy of a Photon |
|
| In 1900, Max Planck was working on the problem of how the radiation an object emits is related to its temperature. He came up with a formula that agreed very closely with experimental data, but the formula only made sense if he assumed that the energy of a vibrating molecule was quantized--that is, it could only take on certain values. The energy would have to be proportional to the frequency of vibration, and it seemed to come in little "chunks" of the frequency multiplied by a certain constant. This constant came to be known as Planck's constant, or h, and it has the value 6.626x10-34 J x s |

|
|
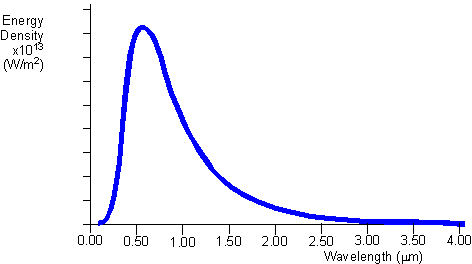
At the time Planck's results could not be explained by current conventional thought regarding light. It was an extremely radical idea to suggest that energy could only come in discrete lumps, even if the lumps were very small. Planck actually didn't realize how revolutionary his work was at the time; he thought he was just fudging the math to come up with the right answer, and was convinced that someone else would come up with a better explanation for his formula. The curve shown sumarizes his findings and is sometimes called the Planck distribution. Based on Planck's work, Einstein proposed that light also delivers its energy in chunks; light would then consist of little particles, or quanta, called photons, each with an energy of Planck's constant times its frequency. Higher-frequency photons have more energy, so they should make the electrons come flying out faster; thus, switching to light with the same intensity but a higher frequency should increase the maximum kinetic energy of the emitted electrons. If you leave the frequency the same but crank up the intensity, more electrons should come out (because there are more photons to hit them), but they won't come out any faster, because each individual photon still has the same energy. |
|
|
|
|
If the frequency is low enough, then none of the photons will have enough energy to
knock an electron out of an atom. So if you use really low-frequency light, you
shouldn't get any electrons, no matter how high the intensity is. Whereas if
you use a high frequency, you should still knock out some electrons even if the
intensity is very low. This was later experimentaly proven by American scientist, R.A. Millikan.. |
|
